Triastek Receives FDA IND Clearance for 3D Printed Medicine for the Treatment of Ulcerative Colitis
Additive Manufacturing
NOVEMBER 30, 2022
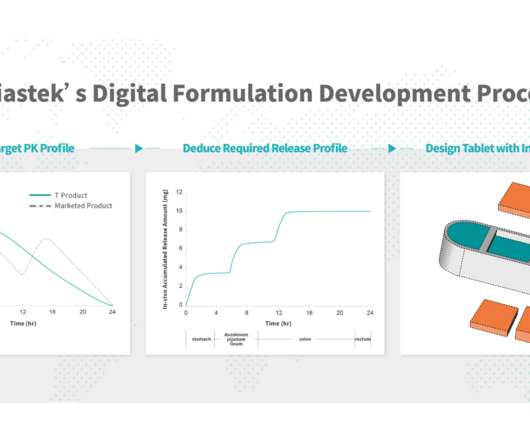
“Delaying drug release and delivering oral dosage forms to the colon is challenging, so T21 offers a promising new option for patients by providing site-specific drug delivery and localized drug effect, mitigating potential side effects from systemic exposure. NANJING, China, Nov. 21, 2022 — Triastek, Inc. ,












































Let's personalize your content